One of nation’s top marijuana experts is at USC. He says pot laws are ‘a political mess’
READ MORE
Medical Marijuana in South Carolina
Read more about the politics, science and businesses surrounding a new push to legalize medical marijuana in South Carolina.
Expand All
When Dr. Prakash Nagarkatti was a little boy and got the flu or the sniffles, his mom often treated him with herbal remedies.
That wasn’t anything unusual in his hometown of Dharwaar, now Dharwad, India. Herbal, or Ayurvedic, medicine has been part of Indian culture for 3,000 years.
The science fascinated Nagarkatti, who went on to earn bachelor’s degrees in both botany and chemistry and a master’s degree in microbiology at Karnatak University in India, then a doctorate degree in immunology at India’s Jiwaji University.
“I was just showing off,” he said of his multiple degrees, laughing.
Today, Nagarkatti is vice president for research at the University of South Carolina, a post he took over in 2011 from now USC President Harris Pastides. He oversees a $250-plus million research enterprise across all USC campuses, and serves as director of the National Institute of Health’s Centers of Research Excellence in Inflammatory and Autoimmune Diseases, among other titles.
Although he presides over all research at South Carolina’s flagship university, his personal research centers on how natural products like red wine, red grapes, green vegetables and ginseng affect the immune system. And for 20 years, Nagarkatti has been one of the nation’s leading researchers into possible medical uses for cannabis, first at Virginia Commonwealth University and now at USC.
That puts him at the forefront of the national debate about whether marijuana has any medical value. The debate has landed in South Carolina, where the state Legislature grapples with a proposal called the Compassionate Care Act to legalize marijuana for medical purposes.
Some research by Nagarkatti and others indicate marijuana contains medical benefits. But researchers, along with law enforcement officials and doctors, are frustrated by what they say is inadequate science and their limited ability to get answers.
Through his work, USC has already patented a U.S. Food and Drug Administration-approved process for treating autoimmune hepatitis with cannabidiol, called CBD, which is derived from hemp, the non-psychoactive cousin of marijuana. Nagarkatti’s team also experiments with tetrahydrocannabino, or THC, the compound in marijuana that gets you high.
(Nagarkatti’s team keeps its THC in tiny vials locked in a refrigerator at an undisclosed location at USC. DEA agents show up regularly and randomly to check on the supply.)
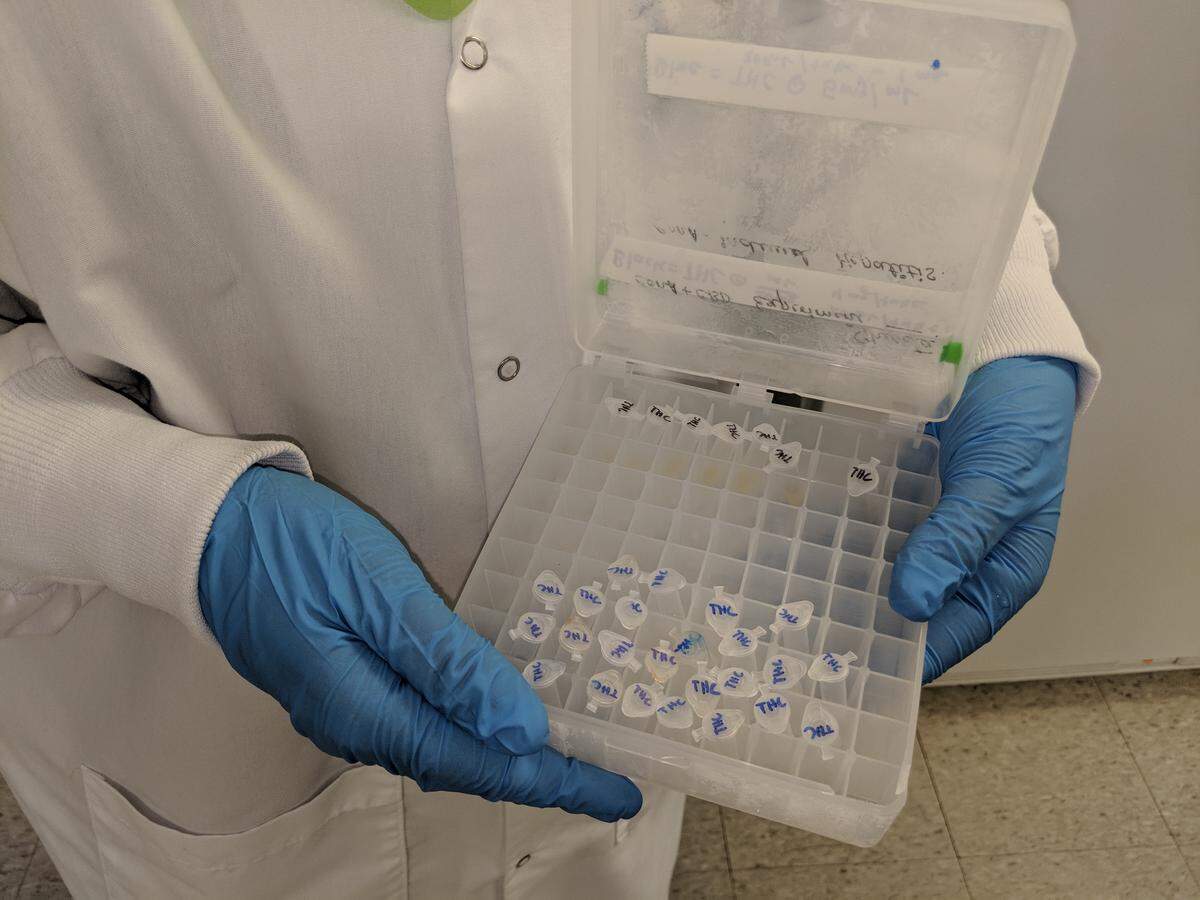
But Nagarkatti and others are frustrated by the limits placed on their research.
The U.S. Justice Department, through the Drug Enforcement Administration, has declared that marijuana is a Schedule 1 drug — the same classification as heroin and LSD. The DEA considers pot, like those drugs, to have no medical value and a high potential for abuse.
So Nagarkatti’s grants — which are administered by the NIH’s National Institute on Drug Abuse — are intended to only explore marijuana’s detrimental effects and not its benefits. Specifically, his research examines the effects of marijuana and its byproducts on the immune system.
However, in looking for the problems caused by marijuana, he and his team are finding that hemp and marijuana derivatives can help with maladies as diverse as battling cancer and obesity to combating deadly bacteria.
“You can’t ignore the science,” Nagarkatti said.
Others have also discovered benefits. In a 1996 study, researchers administered very high doses of THC into mice and rats five times a week for more than two years. They found those rats lived longer and had reduced incidences of several types of cancer.
The FDA itself has acknowledged at least one benefit. Last June, the agency approved a drug from marijuana — Epidiolex, developed in Great Britain — to treat two rare forms of epilepsy, Lennox-Gastaut syndrome and Dravet syndrome, in small children.
“It’s the irony of the whole thing ,“ Nagarkatti said. The DEA and the FDA “are just contradicting themselves.”
‘We need more research’
Whether or not marijuana is “medicine” is central to the state’s debate of the Compassionate Care Act. The bill would allow a doctor to authorize a patient to purchase up to two ounces of pot or an equivalent derivative — oil, cream, vapor, edible, etc., — every two weeks.
The bill’s proponents are convinced medical marijuana can help with a wide range of debilitating conditions from cancer to Post Traumatic Stress Disorder to chronic pain. They also say that legalizing marijuana for medical purposes would help alleviate the opioid crisis by moving patients from pills to pot.
The bill is scheduled to be debated by the Senate Medical Affairs Committee next week.
See highlights of the legislation here.
But opponents, primarily the State Law Enforcement Division chief and the state Medical Association, insist there’s not enough science to justify passing the law.
“SLED is in favor of any marijuana derivative as long as it’s regulated by the FDA, prescribed by a medical physician and dispensed by a legitimate pharmacy,” he said.
Keel, who began his career at 19 as an undercover narcotics agent at a high school, fears a “Pandora’s box of unintended consequences” with legalizing the weed. His pulpit is that it is not recognized by the federal government as medicine and is in fact illegal.
But he adds that he has sympathy for people suffering debilitating illnesses and who may benefit from cannabis-derived medicines. Still, he has a higher duty to protect South Carolinians from the many and varied consequences of illegal drug use.
A SLED scientist says more study is needed to determine if marijuana is medicine.
“We don’t know the effects of long-term high THC use,” C. Todd Hughey, SLED’s laboratory director, said . “We need more research.”
Breaking the law
U.S. scientists haven’t been able to show the FDA that marijuana has medical value because there hasn’t been any large-scale clinical trials on humans. And there hasn’t been any large scale clinical trials because federal grants to study its benefits are not available, and obtaining legal pot can be a bureaucratic nightmare.
Even obtaining a Schedule I drug to research its detriments requires the approval of a posse of federal agencies, among them the DEA, the National Institutes of Health, the National Institute on Drug Abuse and the FDA.
And there is only one place in the entire United States where marijuana is grown and distributed legally: The University of Mississippi.
Ole Miss is home to the National Center for Natural Products Research, which grows cannabis to be distributed to researchers across the country. It’s the only pot farm in the nation that has the federal government’s blessing.
But, you ask: What about the 33 states, District of Columbia, Guam and Puerto Rico that have legalized marijuana for medical use? Or what about the 10 states who have legalized it for recreational use?
They are all breaking federal law.
Pot is still technically illegal at the federal level. And high-flight cannabis researchers like Nagarkatti aren’t going to monkey around with the federal government, which funds their research, OKs their patents and approves any drugs that might emerge. So they follow the federal rules for researching marijuana.
“It’s just a political mess no one wants to fix,” Nagarkatti said.
‘It starts with Schedule 1’
Along with evidence that marijuana has medical benefits, research also has uncovered downsides. Both sets of evidence are found in the same report.
In 2017, the National Academies of Sciences, Engineering and Medicine reviewed 10,000 scientific abstracts published since 1999 and reached nearly 100 conclusions.
An academy committee noted that studies indicate cannabis was effective in reducing pain, improving muscle-related spasms and preventing nausea and vomiting, among other benefits.
But it also noted that in states that have legalized pot, there is increased instances of drugged driving, and the increased risk of unintentional cannabis overdoses among children. There is also some evidence that smoking weed inhibits brain development in children and could cause a heart attack.
Because of the uncertainty, the S.C. Medical Association opposes the Compassionate Care Act, saying doctors would be placed in the position of recommending a treatment not approved by the FDA.
“A review of the anecdotal studies contains some evidence there could be therapeutic uses for some marijuana compounds,” said association President March Seabrook. “It is for this reason we support rescheduling marijuana so the appropriate controlled clinical trials can be performed. Only that process could make marijuana compounds to be considered medicine, and the bill before the legislature seeks to bypass that critical process entirely.”
Keri Holmes-Maybank, a clinical researcher at the Medical University of South Carolina, agrees that leaf marijuana or derivatives not approved by the FDA should not be made available to the public.
“We just don’t know enough,” she said. “We need study and we need the federal government and the private sector to make that happen. . . . that starts with the Schedule 1 and the FDA. It’s really prohibitive.”
Should S.C.’s Compassionate Care Act pass and be signed by Gov. Henry McMaster, Nagarkatti’s research stands to benefit. The bill would give 10 percent of the revenues generated above the state’s cost to research, with the money evenly split between USC School of Medicine and MUSC.
USC has been studying marijuana since 2005, Nagarkatti said, and the additional state funding would help kick start the clinical trials most researchers and related organizations advocate.
“Because it is difficult to secure NIH funding to test the beneficial effects of marijuana, the state funds would be very helpful to pursue such research,” he said. “Also, clinical trials can be performed at MUSC and thus, the collaborations would be very fruitful.”
This story was originally published April 4, 2019 at 5:00 AM.